A Careful Interpretation of the Audiometry may contribute to the Diagnosis of Alport Syndrome: A Pilot Study
Claudio Alves Andrade Cardoso#, Danilo Euclides Fernandes#*, Michelle Tiveron Passos Riguetti, Gianna Mastroianni Kirsztajn
Universidade Federal de Sao Paulo (UNIFESP), Sao Paulo - SP, 04021-001, Brazil
Abstract
INTRODUCTION: Hearing loss is a typical finding in Alport syndrome (sAlport) caused by a genetic defect in the type IV collagen synthesis. Depending on the criteria, these individuals may be diagnosed with normal hearing.
OBJECTIVE: To compare the prevalence of hearing loss according to different criteria – World Health Organization, WHO; Global Burden Disease, GBD and Clark – in patients with sAlport and segmental and focal glomeruloesclerosis (FSGS).
MATERIALS AND METHODS: This is a cross-sectional pilot study. Pure-tone audiometry was carried out in patients with sAlport and FSGS (glomerulopathy which was selected as a control group).
RESULTS: We assessed 13 patients (6 with sAlport and 7 with FSGS). Under the WHO criteria, no patient had hearing loss. The prevalence of hearing loss was similar according to GBD criteria (16.67% and 14.29% in the sAlport and FSGS groups, respectively). Clark's criteria, instead, revealed a higher prevalence of hearing loss in the sAlport group (66.67%) vs. FSGS (28.57%).
CONCLUSION: The prevalence of hearing loss in the sAlport group varied depending on the criteria (from nonexistent to 67%). We consider that a critical evaluation of the hearing thresholds may help physicians to early detect minimal hearing impairment even though the report says, “normal hearing”.
INTRODUCTION
Despite the different pathways of embryonic development, kidneys and ears share similar mechanisms that can influence both organogenesis1–3. Alport syndrome (sAlport), for example, is known for its phenotype of renal, auditory and ocular involvement4 and affects 1/10.000 individuals. The pathophysiology of this disease lay on a genetic defect in type IV collagen synthesis that interferes with the glomerular filtration barrier4 and degenerates cochlear vascular stria5.
Usually, the sAlport presents with sensorineural hearing loss6 and renal loss of function4. The early onset – young adulthood6 or at childhood7 – and the family history of renal/hearing impairment may suggest sAlport. Depending on the variants, some individuals present only with renal failure, hardening sAlport diagnosis8,9.
The World Health Organization (WHO) suggests that the best ear mean of thresholds (500, 1000, 2000 and 4000 Hz) should not exceed 25 dB. If so, the patient has hearing loss10. In many studies the criteria for hearing loss remain unclear9,11-15, others use WHO’s recommendations8. Both cases can “hide” milder hearing losses.
Because of some incongruencies at setting a cut-off for hearing threshold, we compared the prevalence of hearing loss through different criteria10,16,17 in two groups of patients with glomerulopathies – one group with sAlport and the other, the control group, with segmental and focal glomerulosclerosis (FSGS), which share many clinical characteristics.
MATERIALS AND METHODS
This is a cross-sectional, single-center, pilot study developed at the Outpatient Clinic of Glomerulonephritis from Universidade Federal de São Paulo (UNIFESP), Brazil. This protocol was approved by the Research Ethics Committee of UNIFESP (CAAE 96087918.9.0000.5505) and developed according to the principles of the Helsinki Declaration. All participants provided their written consent.
We included patients younger than 65 years-old that had biopsy-proven kidney disease (optical microscopy, immunofluorescence and, electron microscopy in sAlport). The individuals of the sAlport group presented familial hematuria and typical renal lesion of Alport disease at electron microscopy. Kidney biopsy also confirmed FSGS in the control group. Patients who had other possible causes of hearing loss (exposure to occupational noise, history of repeated otitis media or otosclerosis and type B tympanometry) were excluded.
Demographic and clinical data (weight, height, BMI, gender, age, ethnicity, family history of hearing loss and/or renal disease, high blood pressure, serum creatinine, 24-hour proteinuria and presence of hematuria) were obtained from the patients' charts. Audiometry was performed by an audiologist and included external auditory canal examination and tonal and vocal audiometry.
We used the quadritonal mean (Mq) of the auditory thresholds for the frequencies of 0.5, 1, 2 and 4 kHz to determine hearing loss according to three criteria: WHO (Mq > 25 dB)10, Global Burden Disease (Mq > 20 dB)17 and Clark (Mq > 15 dB)16.
The data were organized in Microsoft Excel for Mac, version 16.35 and analyzed in IBM® SPSS Statistics software, version 26. We present summarized results through relative and absolute frequencies (nominal variables) and mean and standard deviation (numerical variables). Inferential analyses were performed using Pearson's Chi-square test and Wilcoxon's test. We also compared Clark [10] and WHO [12] criteria regarding sensitivity and specificity (ROC curve). We set alpha= 0.05 and 95% confidence interval.
RESULTS
We analyzed 13 patients (sAlport=6 and FSGS=7). Most of the individuals were women (69.23%), adults (42.69 ± 18.35 years old), other ethnicities than white (38.46%), and normal- weighted (body mass index = 25.18 ± 2.84). The sAlport group (37.17 ± 19.41) were younger than the FSGS group (47.43 ± 17.40) (p=0.33). Most of them (84.62%) had a family history of chronic kidney disease (CKD), and some (38.46%) had a family history of hearing loss. sAlport group presented a higher frequency of family history for both CKD (100%) and hearing loss (66.67%). Family history of CKD among the FSGS group was (71.43%) (Table 1). None of the individuals were on renal replacement therapy.
Although most patients had high blood pressure (69.23%), we observed that FSGS group presented higher systolic pressure, serum creatinine 1.4 (± 0.62) mg/dL, and 24-hour proteinuria 1.5 (± 1.87) g. The sAlport group showed hematuria, instead (Table 1).
The prevalence of hearing loss varied according to the criteria: no hearing loss (WHO), similar prevalence (GBD; 16.67% in the sAlport group and 14.29% in the FSGS group), and different prevalence (Clark; 66.67% in the sAlport group and 28.57% in the FSGS group) (Table 1). In sAlport group, two individuals presented mild hearing loss: a man and a woman.
Figure 1 shows the thresholds, between 250 and 8000 Hz, on the right and left ears of each group.
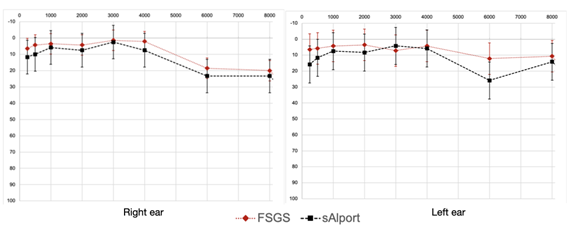
Figure 1: Mean and standard deviation (error bars) of the pure-tone audiometry from 250 to 8000 Hz in FSGS (in red) and sAlport group (in black). Abbreviations: FSGS, focal and segmental glomerulosclerosis; sAlport, Alport syndrome.
Table 1: Demographic characteristics and comparisons between the two groups of patients (with and without Alport syndrome).
|
sAlport |
(n = 6) |
FSGS |
(n = 7) |
p-value |
|||
|
Weight |
62.80 |
(10.11) |
69.76 |
(5.80) |
0.149a |
||
|
Height |
1.63 |
(0.15) |
1.63 |
(0.05) |
0.968a |
||
|
BMI |
23.74 |
(2.95) |
26.42 |
(2.25) |
0.9a |
||
|
Sex |
|||||||
|
Female |
4 |
(66.67%) |
5 |
(71.43%) |
1.00b |
||
|
Male |
2 |
(33.33%) |
2 |
(28.57%) |
|||
|
Age |
37.17 |
(19.41) |
47.43 |
(17.40) |
0.336a |
||
|
Ethnicity |
|||||||
|
Not reported |
1 |
(16.67%) |
2 |
(28.57%) |
0.69b |
||
|
White |
0 |
(0.00%) |
4 |
(57.14%) |
|||
|
Other ethnicities than white |
5 |
(83.33%) |
1 |
(14.29%) |
|||
|
Family history of CKD |
6 |
(100.00%) |
5 |
(71.43%) |
0.462b |
||
|
Family history of HL |
4 |
(66.67%) |
1 |
(14.29%) |
0.103b |
||
|
Reports HL |
1 |
(16.67%) |
0 |
(0.00%) |
0.462b |
||
|
Chronic hypertension |
3 |
(50.00%) |
6 |
(85.71%) |
0.266b |
||
|
SBP, in mmHg, mean (sd) |
116.7 |
(6.06) |
120.7 |
(13.67) |
0.518a |
||
|
DBP, in mmHg, mean (sd) |
79.2 |
(12.01) |
77.1 |
(9.51) |
0.741a |
||
|
Serum creatinine |
0.8 |
(0.07) |
1.4 |
(0.62) |
0.057a |
||
|
24-hour Proteinuria |
0.2 |
(0.32) |
1.5 |
(1.87) |
0.105a |
||
|
Hematuria |
|||||||
|
< 100.000 |
2 |
(33.33%) |
7 |
(100.00%) |
0.021b |
||
|
> 100.000 |
4 |
(66.67%) |
0 |
(0.00%) |
|||
|
Hearings loss, WHO, 1991 (RE+LE)c |
0 |
(0.00%) |
0 |
(0.00%) |
- |
||
|
Hearings loss, GBD, 2013 (RE+LE)d |
1 |
(16.67%) |
1 |
(14.29%) |
0.759b |
||
|
Hearings loss, Clark, 1981 (RE+LE)e |
4 |
(66.67%) |
|
2 |
(28.57%) |
|
0.559b |
a Wilcoxon Test
b Pearson's Chi-Square Test
cWHO, 1991 [9]: quadritonal mean (0,5, 1, 2, 4 kHz) > 25
dGBD, 2013 [11]: quadritonal mean (0,5, 1, 2, 4 kHz) >20
eClark, 1981 [10]: quadritonal mean (0,5, 1, 2, 4 kHz) > 16
Abbreviations: BMI, body mass index; CKD, chronic renal disease; HL, hearing loss; SBP, systolic blood pressure; DBP, diastolic blood pressure; sd, standard deviation; n, sample; WHO, World Health Organization; GBM, Global Burden Disease; RE, right ear; LE, left ear.
Table 2: Comparison between sensitivity and specificity of different hearing loss criteria by the quadratic mean.
|
|
AUC |
AUC difference |
p-value |
(95% CI) |
|
Minimum hearing loss (Clark, 1981) |
0.595 |
0.095 |
0.293 |
(-0.07; 0.26) |
|
Hearing loss (WHO, 1991) |
0.500 |
Abbreviations: WHO, World Health Organization; AUC, area-under-the-curve; CI, confidence interval.
Clark’s criteria (Mq > 15 dB) detected hearing loss with a higher sensitivity than the WHO (Mq > 25 dB). Our comparison (ROC curve) showed statistical significance between the area-under-the-curve of them (AUC difference = 0.095; p = 0.293, 95% CI -0.07; 0.26) (Table 2).
Table A (supplementary material) shows that the higher the cut-off for hearing loss, the higher specificity of the criteria. Instead, sensitivity reached 0% around 25 dB, which remains the normal hearing threshold according to WHO.
Table A: Sensitivity and specificity values for quadritonal averages.
|
|
dB |
Sensitivity |
Specificity |
|
Quadritonal mean |
-9 |
1.00 |
0.00 |
|
-7 |
1.00 |
0.07 |
|
|
-5 |
0.92 |
0.07 |
|
|
-3 |
0.92 |
0.14 |
|
|
-1 |
0.92 |
0.21 |
|
|
1 |
0.67 |
0.29 |
|
|
2 |
0.67 |
0.50 |
|
|
3 |
0.67 |
0.64 |
|
|
5 |
0.50 |
0.79 |
|
|
7 |
0.42 |
0.79 |
|
|
8 |
0.42 |
0.86 |
|
|
13 |
0.33 |
0.86 |
|
|
18 |
0.25 |
0.93 |
|
|
22 |
0.08 |
0.93 |
|
|
26 |
0.00 |
0.93 |
|
|
29 |
0.00 |
1.00 |
Abbreviations: dB, decibel.
DISCUSSION
This is a pilot study that investigated the prevalence of sensorineural hearing loss in patients with sAlport, according to three different criteria: Clark16, GBD17 and WHO10. We also compared the sensitivity and specificity of Clark and WHO criteria and observed that Clark’s criteria (Mq > 15 dB) favors hearing loss diagnosis.
sAlport phenotype remains well known because of its triad: eye, ear and kidney impairment. Their organogenesis seems to be unrelated, but some transcription factors are common to renal and auditory development1-3 and the clinical presentation lays on the structural defect of type IV collagen. More commonly, sAlport is linked to chromosome X (COL4A5 mutation) and 80% of the individuals have this pattern of inheritance. The most frequent phenotype includes a family history of hematuria, CKD, and early-onset hearing impairment7. The diagnosis of renal involvement in the Alport Syndrome is based on electron microscopy typical findings. Hearing loss is described as sensorioneural, bilateral, and symmetrical, even though its degree can vary, which we had already observed13.
Even for those patients who have isolated high frequency hearing loss (30% to 50%), renal impairment is always present. The opposite is not observed, and renal impairment can occur solely. Also, the severity of ear and kidney dysfunction seemed to be unrelated18.
In this study, we used a similar renal group – corticoresistant familial FSGS – to compare its hearing thresholds to the sAlport’s. About 20% of familial FSGS cases present type IV collagen mutations19, which is found in all cases of FSGS we used here.
As we recruited individuals from an outpatient service of glomerulonephritis, none of the subjects were on dialysis at the time we conducted this protocol.
FSGS and sAlport groups were comparable in BMI and gender, but not in age because sAlport group was considerably younger than the FSGS group, possibly because the patients were recruited from an outpatient clinic that assists individuals over 12 years old. But, despite their age, sAlport group presented a higher prevalence of hearing loss by Clark’s and GBD’s criteria, which was unlikely considering their age. They, also, presented a higher frequency of family history for CKD and hearing loss, which were expected. We avoided the confounding factor of age on hearing loss by not including individuals older than 65 years old, which may be affected by presbycusis. Even younger, our sAlport group did not meet the WHO’s criteria for hearing loss7, which may be found among individuals who are older than 40 years old. Additionally, the Alport Syndrome patients we included did not complain of any clinical hearing loss.
The WHO’s criteria (Mq > 25 dB) is widely used in Audiology and is based on expert opinion to suggest its cut-off. More recently, they suggested that a functional loss should be considered in the borderline cases of hearing loss (around 20 dB of quadritonal mean)10,16,17,20. With our patients, we observed that GBD’s criteria17 fits better in identifying milder grades of hearing loss, as already shown21. Clark’s criteria16, instead, differentiated the two groups we compared. At Clark’s cut-off, patients may not complain about hearing, but they may be aware of difficulties in listening-in-noise situations, which highlights a mild hearing impairment. Despite hearing difficulties, these patients can complain about hearing discomfort or tinnitus, which can worsen their quality of life22. Here, we observed that a lower cut-off facilitates the hearing assessment when it comes to sAlport investigation.
The main limitations of our study are the small sample size and the number of patients included in the FSGS Group. Although sample size limits our statistical analyses, it does not invalidate the discussion of the hearing loss criteria we propose in this pilot study. Also, our study highlights the highly variable presentations of pure-tone audiometry in Alport Syndrome. The absence of hearing complaints made it difficult for us to expand the FSGS group, because these patients were unmotivated to participate in a hearing investigation, which they considered unnecessary.
CONCLUSION
Depending on the criteria, audiometry can hide mild hearing losses. We consider that a critical evaluation of the hearing thresholds may help physicians to early detect minimal hearing impairment even though the report says, “normal hearing”, especially in a group of patients without end-stage renal disease.
References
- Lin F, Hiesberger T, Cordes K, et al. Kidney-specific inactivation of the KIF3A subunit of kinesin-II inhibits renal ciliogenesis and produces polycystic kidney disease. Proc Natl Acad Sci USA. 2003; 100(9):5286–5291.
- Torban E, Goodyer P. The kidney and ear: emerging parallel functions. Annu Rev Med. 2009; 60:339–353.
- Izzedine H, Tankere F, Launay-Vacher V, Deray G. Ear and kidney syndromes: molecular versus clinical approach. Kidney Int. 2004; 65(2):369–385.
- Kruegel J, Rubel D, Gross O. Alport syndrome--insights from basic and clinical research. Nat Rev Nephrol. 2013; 9(3):170–178.
- Gregg JB, Becker SF. Concomitant Progressive Deafness, Chronic Nephritis, and Ocular Lens Disease. Archives of Ophthalmology. 1963; 69(3):293–299.
- Nozu K, Nakanishi K, Abe Y, et al. A review of clinical characteristics and genetic backgrounds in Alport syndrome. Clin Exp Nephrol. 2019; 23(2):158–168.
- Lee JM, Nozu K, Choi DE, Kang HG, Ha I-S, Cheong HI. Features of Autosomal Recessive Alport Syndrome: A Systematic Review. J Clin Med. 2019; 8(2).
- Zhang X, Zhang Y, Zhang Y, et al. X-linked Alport syndrome: pathogenic variant features and further auditory genotype-phenotype correlations in males. Orphanet J Rare Dis. 2018; 13(1):229.
- Storey H, Savige J, Sivakumar V, Abbs S, Flinter FA. COL4A3/COL4A4 Mutations and Features in Individuals with Autosomal Recessive Alport Syndrome. JASN. 2013; 24(12):1945–1954.
- Deafness IWG on P of, Hearing Impairment Programme Planning (1991: Geneva S, Deafness WHOP for the P of, Impairment H. Report of the Informal Working Group on Prevention of Deafness and Hearing Impairment Programme Planning, Geneva, 18-21 June 1991. World Health Organization; 1991.
- Savige J, Storey H, Il Cheong H, et al. X-Linked and Autosomal Recessive Alport Syndrome: Pathogenic Variant Features and Further Genotype-Phenotype Correlations. Singh SR, editor. PLoS ONE. 2016; 11(9):e0161802.
- Chugh KS, Sakhuja V, Agarwal A, et al. Hereditary nephritis (Alport’s syndrome)—clinical profile and inheritance in 28 kindreds. Nephrology Dialysis Transplantation. 1993; 8(8):690–695.
- Viveiros CM, Pereira LD, Kirsztajn GM. Auditory perception in Alport’s syndrome. Braz J Otorhinolaryngol. 2006; 72(6):811–816.
- Sirimanna KS, France E, Stephens SD. Alport’s syndrome: can carriers be identified by audiometry? Clin Otolaryngol Allied Sci. 1995; 20(2):158–163.
- Zakzouk SM, Fadle KA, Al Anazy FH. Familial hereditary progressive sensorineural hearing loss among Saudi population. International Journal of Pediatric Otorhinolaryngology. 1995; 32(3):247–255.
- Clark JG. Uses and abuses of hearing loss classification. ASHA. 1981; 23(7):493–500.
- Stevens G, Flaxman S, Brunskill E, Mascarenhas M, Mathers CD, Finucane M. Global and regional hearing impairment prevalence: an analysis of 42 studies in 29 countries. European Journal of Public Health. 2013; 23(1):146–152.
- Yu AS. Brenner & rector’s the kidney. 11th edition. St. Louis, MO: Elsevier; 2019.
- Rheault MN, Savige J, Randles MJ, et al. The importance of clinician, patient and researcher collaborations in Alport syndrome. Pediatr Nephrol. 2020; 35(5):733–742.
- Olusanya BO, Davis AC, Hoffman HJ. Hearing loss grades and the International classification of functioning, disability and health. Bull World Health Organ. 2019; 97(10):725–728.
- Alves FRA, Andrade Quintanilha Ribeiro F de. Clinical data and hearing of individuals with Alport syndrome. Brazilian Journal of Otorhinolaryngology. 2008; 74(6):807–814.
- Choi JE, Ahn J, Park HW, Baek S-Y, Kim S, Moon IJ. Prevalence of minimal hearing loss in South Korea. PLoS One [Internet]. 2017 [cited 2020 May 13]; 12(2). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5308612/
