Postorgasmic Illness Syndrome: What do we know so far?
Tan Vu Le1, Hoang Minh Tue Nguyen1, Wayne J.G. Hellstrom1*
1Tulane University School of Medicine, Department of Urology, New Orleans LA, USA
Abstract
Post-orgasmic illness syndrome (POIS) is rare condition that is characterized by transient flu-like symptoms and cognition disorders that occur shortly after ejaculation and last for 2-7 days. There are about 50 cases of POIS in the literature. The prevalence and incidence of POIS are still unknown due to a paucity of studies. The exact pathogenesis of POIS remains unknown; the most acceptable hypothesis is an autoimmune/allergic process. We made a literature search via PubMed for publications from 2002 to 2018 with the “post orgasmic illness syndrome” medical subject heading term to analyze current data regarding symptoms, burden, pathophysiology, and to discuss potential management options for POIS. POIS is diagnosed by five preliminary diagnostic criteria. It is categorized into two types: primary and secondary. There is the concomitance between POIS and PE. The autoimmune/allergy hypothesis is the most accepted hypothesis explaining POIS pathogenesis. A competing hypothesis involves a disorder of endogenous μ-opioid receptors. Patients with POIS have been symptomatically treated with antihistamines, selective serotonin reuptake inhibitors, and benzodiazepines. A trial of hyposensitization therapy with autologous semen was successful. A trial of nonsteroidal anti-inflammatory medication helped in a single case report but failed to successfully treat other patients with POIS.
Introduction
Post-orgasmic illness syndrome (POIS) is a rare condition that was first described by Waldinger and Schweitzer in 20021. Two men in their study suffered from exhaustion, flulike symptoms (intense warmth, perspiration, headache, burning eyes, sore throat, general myalgia), erythema of the skin (itching, pimples), urinary hesitation, disordered stooling, and mental disturbances (reduced concentration and agitation). Notably, all of these symptoms occurred shortly after ejaculation and lasted to 2 - 7 days1.
In an effort to limit the symptomatology, most patients suffering from POIS resort to reducing sexual activity or abstinence1-8. The physical and psychological effects of POIS significantly affect the quality of life of both patients and their partners1-12.
Approximately, 50 cases have been recorded in the literature since 20024,5. The prevalence and incidence of POIS are unknown owing to a paucity of studies5,12. Most of the studies are case reports. POIS is underdiagnosed and underreported, although there are an increasing number of self-reported cases of POIS in Internet forums5,6.
Various theories have been postulated on the pathophysiology of POIS2,4,5. The most pervasive explanation is attributed to Waldinger and Schweitzer who hypothesized an immune-modulated mechanism as the underlying etiology4.
This mini-review seeks to provide an update on the current literature on POIS. The authors also aim to provide updated information regarding the pathophysiology of POIS and to discuss potential management options.
Methods
We conducted a 2002 to 2018 literature search using PubMed. The following medical subject heading terms were used: “orgasm illness”, “post-orgasmic”, “post-orgasmic illness syndrome.” Inclusion criteria for article selection were studies (of any design) on post-orgasmic illness in men, and publication in a peer-reviewed journal. The reference lists of identified publications were reviewed manually for additional relevant articles. Manuscripts written in languages other than English were accepted.
Clinical Presentation
In 2011, Waldinger et al. reported the clinical characteristics of 45 Dutch patients with POIS and were able to define five diagnostic criteria3 (Table 1).
Table 1: Five preliminary diagnostic criteria of POIS3
| 1. One or more of these following symptoms: - General: Extreme fatigue, exhaustion, palpitations, anomic aphasia, incoherent speech, dysarthria, concentration difficulties, irritability, hyperacusis, photophobia, depressed mood - Flu-like: Fever, extreme warmth, perspiration, chills, prodrome, cold intolerance - Head: Headache, fogginess, heaviness in the head - Eyes: Burning, conjunctival injection, blurry vision, eye pain, watery discharge, eye irritation and itchiness - Nose: Nasal congestion, rhinorrhea, sneezing - Throat: Dirty taste in mouth, dry mouth, sore throat, tickling cough, hoarse voice - Muscle: Muscle tension in the back or neck, muscle weakness and pain, heaviness in the legs, muscle stiffness |
| 2. Symptoms occur seconds to a few hours after ejaculation |
| 3. Symptoms occur always or nearly always, i.e., in more than 90% of ejaculation events |
| 4. Symptoms last for about 2 to 7 days |
| 5. Symptoms then disappear spontaneously |
They proposed that the presentation of POIS was highly variable; however, the most common symptoms were: concentration difficulties, extreme fatigue/ exhaustion and fever/ body warmth/ perspiration/ shivering3.
Furthermore, POIS symptoms started within 30 minutes of ejaculation in 87% of afflicted men in the Waldinger et al. study3. Notably, of the 33 men (73%) with a partner, the intercourse frequency was 1.04 ± 1.00 times per week; three men had decided to abstain completely from intercourse3. Eight of these 33 men reported an intercourse frequency of once in two to six months3. Of the males older than 30 years old without a partner, six men refrained from masturbation or intercourse as much as possible3. Other authors recorded similar reduced sexual activity frequency in POIS patients2,5,7,15. All of these represent the consequences of POIS (Figure 1).
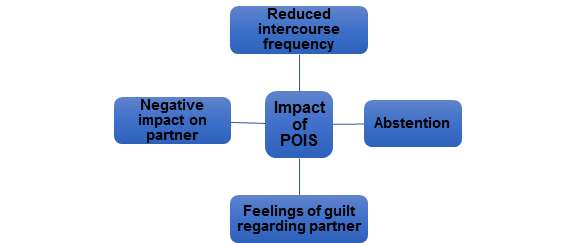
Figure 1. Impacts of POIS on afflicted patients and their partners2,3,5-7,13
It should be emphasized that POIS not only affected men but also their partners13,14. Nearly all men were concerned about the relationship with their sexual partner and expressed feelings of guilt for having the disorder3. In the series of 33 couples in the Waldinger et al. study, there were three divorces during follow-up; two of which were due to patients’ strategy of abstinence or avoidance of sexual activity. On the other hand, two initially single males married female partners who accepted the negative sexual consequences of their disorder3. The majority of case reports emphasized that POIS caused a negative impact on the partners2,6,7.
The patients with POIS should undergo a complete medical history that includes existing allergies, sexually transmitted diseases, and any history of prostatitis. In addition, a neuropsychiatric interview regarding possible neurological or psychiatric disorders, as well as a sexual function interview with special attention to ejaculatory disorders and their partner relationship should both be recorded2,3,16,18. Patients should be carefully examined and screened for other diseases that have similar symptoms to POIS. This should include recording the duration of symptoms, an assessment of bothersome symptoms and a detailed lower urinary tract symptoms review. A digital rectal examination (DRE) needs to be performed after a midstream urine sample has been collected for urine dipstick, microscopy, and culture2,3,16,18. The males who are clinically diagnosed as having POIS undergo routine tests, including full blood count, electrolytes, creatinine, liver function tests and hormonal laboratory (follicle stimulating hormone, luteinizing hormone, prolactin, testosterone). Magnetic resonance imaging brain scans may be needed to rule out abnormal conditions in the brain that might lead to a headache after ejaculation. Notably, no condition associated with ejaculation has been recorded in cases of POIS through pathology history, examination findings or laboratory tests2,3,5,6,15,16,18.
A thorough history and examination are both important in making a differential diagnosis, especially prostatitis or chronic pelvic pain, which can manifest similarly as flu-like symptoms, weakness and pain of the muscle localized to the perineum or suprapubic area. Nevertheless, symptoms of prostatitis typically last longer than those of POIS (2-7 days). In addition, pain during or after ejaculation and lower urinary tract symptoms are the most prominent, and bothersome features in prostatitis patients. A DRE may reveal a nodule or tenderness and pain in the prostate8,9,10.
Primary POIS and Secondary POIS
Based on the study of 45 males who fulfilled the aforementioned five criteria, Waldinger et al categorized two types of POIS: a primary type in which POIS becomes manifest from the first ejaculation in puberty or adolescence, and a secondary type in which POIS manifests later in life3. Among 45 males in their study, 49% had the primary type, whereas 51% had the secondary type3. According to this classification, there were 5 primary and 4 secondary POIS patients who have been described in the literature by other authors2,4,5,7,15-18.
POIS and PE
Waldinger et al commented that one POIS patient in his study suffered from lifelong (PE), and another had acquired PE1. Among 45 POIS cases in the Waldinger et al. study, 56% of men reported lifelong PE with an intravaginal ejaculation latency time of less than one minute3. The authors estimated that the relative risk of men with POIS was 22.4-fold higher to develop PE than for healthy individuals3. Jiang et al., Shigeta et al. and Bignami B. et al have also reported lifelong PE in their three POIS patients5,7,19. It may be speculated that PE in this group of men is induced by forced abstinence and the low frequency of sexual activity3. Another reason was these men also had to cope with reducing ejaculation frequency as much as possible even when the urge to have sex and intimacy is normal or even strong as the consequences of an ejaculation would be a disturbance to their life and work14.
Pathophysiology
Different hypotheses have been postulated on the pathophysiology of POIS, including an immunological phenomenon, an opioid-like withdrawal or a neuro-endocrine response2-5,15 (Table 2). Nevertheless, the most predominant explanation is attributed to Waldinger et al., who hypothesized an immune modulated mechanism as the underlying etiology3. This theory was supported by a study of skin-prick tests (SPT). After performing this test in 33 patients with POIS using extremely diluted samples of their own semen (1:40,000), the authors reported that 88% had positive reactions3. Limitations of this study were the lack of performing the SPT with autologous semen in an age-matched control group of healthy men and the lack of measuring the serum specific immunoglobin E (IgE) in the POIS patients. To address this limitation of the Waldinger et al. study, Kim et al. confirmed the existence of serum semen-specific IgE in their patient with POIS15.
In contrast to the autoimmune/allergy hypothesis, Jiang et al. proposed that an IgE-mediated semen allergy in POIS-afflicted men might not adequately account for the symptomatology5. They performed a SPT and other intracutaneous tests in a patient with POIS and three healthy controls5. The results countered the immune-mediated hypothesis; three healthy men without POIS showed positive skin test reactions to injection with autologous semen. The authors suggested that there was no evidence of semen-specific IgE antibodies in men with POIS and positive skin reactions to autologous semen5. Instead, they compared the symptoms of POIS to opioid withdrawal, which include similar physical and psychological manifestations5,20.
Ashby and Goldmeier proposed an alternate hypothesis where POIS was driven by a disordered cytokine or neuroendocrine response2. This was supported by the improvement of POIS symptoms in the patient following administration of prophylactic diclofenac, a nonsteroidal anti-inflammatory drug (NSAID)2.
Management
Currently, there is no specific treatment for POIS, in part, due to varying pathophysiological hypothesis (Table 2). Patients with POIS-like symptoms have been treated with antihistamines, prednisone, benzodiazepines, and tramadol1,17,19. Selective serotonin reuptake inhibitors (SSRIs) may be considered in the concomitant treatment of POIS and PE19.
Table 2: Hypothesis and management of POIS
| Year | Authors | Numbers of cases | Hypothesis | Treatment recommendation |
|---|---|---|---|---|
| 2011 | Waldinger et al [3,4] | 47 | Immune modulated mechanism | Subcutaneous immunotherapy with autologous semen |
| 2018 | Kim et al [15] | 1 | Intralymphatic immunotherapy with autologous semen | |
| 2010 | Ashby et al [2] | 2 | Disordered cytokine or neuroendocrine response | Symptoms treatment2,17,19: Antihistamines, selective serotonin reuptake inhibitors, and benzodiazepines |
| 2015 | Jiang et al [5] | 1 | Opioid withdrawa | Diclofenac 75mg 1-2 hours prior to sexual activities with orgasm, continue twice daily 24-48 hours |
Considering the successful outcomes of hyposensitization therapy in clinical allergic diseases, Waldinger et al. reported the improvement of POIS symptoms by the hyposensitization treatment with autologous semen in two Dutch patients4. The protocol of treating POIS was developed by Marcus Meinardi3,4. He intensified the hyposensitization by starting with extremely diluted autologous semen and gradually employing higher concentrations of autologous semen4. To modify this treatment, Kim et al. performed intralymphatic immunotherapy with autologous semen instead of subcutaneous delivery on a Korean male with POIS15. This is a novel method of allergen-specific immunotherapy being used in allergic diseases. Using ultrasound guidance and a 25-gauge needle, autologous semen was aseptically injected into an inguinal lymph node at a dilution of 1:40,00015. Then, the concentration was increased by 3-fold, as in a previous study by Waldinger4,15. After the 3rd and 4th injections, the patient complained of flu-like symptoms which persisted for 3 to 4 weeks with an intensity that remained at 50% to 60% 5 days after the 3rd injection and at 60% to 70% 5 days after 4th injection15. Notably, at 8 and 15 months after the first injection, all POIS- related symptoms except sore throat and urinary symptoms were alleviated and their durations were shortened15. The authors recommended that hyposensitization therapy could have therapeutic effects in patients with POIS in whom allergies are a dominant etiologic factor15. Nevertheless, limitations of hyposensitization treatment include a lack of healthy male controls for the autologous semen skin test results. Consequently, its true efficacy remains unconfirmed.
Another successful trial of therapy is a NSAID (diclofenac), which succeeded in reducing symptoms (up to 80% improvement) and allowed the patient in that case report to increase his sexual frequency from twice a month to four times a month2,5,16. However, NSAIDs therapy has failed in other patients, highlighting the need for further investigation into the nature and treatment of POIS. Additionally, numerous anecdotal therapies have been suggested to be efficacious in improving POIS symptoms, including niacin, olive leaf, fenugreek, saw palmetto, and wobenzym6.
Conclusion
POIS is a rare condition and its true prevalence remains unknown. POIS can cause severe distress that can affect both the POIS-afflicted male and his partner. Patients with POIS manifest with a constellation of flu-like and allergic symptoms that commence seconds, minutes, or hours after ejaculation and last for several days. POIS characterized by five criteria and its symptoms can be described by seven clusters. Depending on the time of onset, POIS is classified into primary and secondary categories. Notably, the combination of POIS and lifelong PE is apparent and deserves further attention in future POIS treatment strategies. The exact pathogenesis of POIS remains unknown, but the most acceptable hypothesis is an autoimmune/allergic process. On the other hand, chemical imbalances in the brain similar to opioid withdrawal or dysregulation in cytokine and chemokine response are postulated. Hence, no definitive treatment for POIS has been confirmed, but hyposensitization was successful in three patients4,15. Further research on the reactivity of healthy men to autologous semen, the roles of neurotransmitters in POIS as well as the relationship between POIS and PE need further investigation.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of Interest
All authors claim no conflict of interest regarding the content of this paper.
References
-
- Waldinger MD, Schweitzer DH. Postorgasmic illness syndrome: two cases. J Sex Marital Ther. 2002 May-Jun; 28(3): 251-5.
- Ashby J, Goldmeier D. Postorgasm illness syndrome-a spectrum of illnesses. J Sex Med. 2010; 7: 1976–81.
- Waldinger MD, Meinardi MM, Zwinderman AH, et al. Postorgasmic illness syndrome (POIS) in 45 dutch Caucasian males: Clinical characteristics and evidence for an immunogenic pathogenesis (part 1). J Sex Med. 2011; 8: 1164–70.
- Waldinger MD, Meinardi MM, Schweitzer DH. Hyposensitization therapy with autologous semen in two Dutch caucasian males: beneficial effects in Postorgasmic Illness Syndrome (POIS; Part 2). J Sex Med. 2011 Apr; 8(4): 1171-6.
- Jiang N, Xi G, Li H, et al. Postorgasmic illness syndrome (POIS) in a Chinese man: no proof for IgE-mediated allergy to semen. J Sex Med. 2015 Mar; 12(3): 840-5.
- Daveman. Postorgasmic illness syndrome (POIS). Available at http://www.poiscenter.com. Accessed May 31, 2017.
- Shigeta K, Kikuchi E, Matsushima M, et al. A clinical case of suspected postorgasmic illness syndrome. J Sex Med. 2013; 10(Supp 3): 220.
- Gujadhur R, Aning J. Careful assessment key in managing prostatitis. Practitioner. 2015; 259 (1781): 15-19.
- Rees J, Abrahams M, Doble A. Diagnosis and treatment of chronic bacterial prostatitis and chronic prostatitis/chronic pelvic pain syndrome: a consensus guideline. BJU INT. 2015; 116: 509-525.
- Doiron C, Curtis J. Evaluation of the male with chronic prostatitis/chronic pelvic pain syndrome. Canadian Urological Association Journal. June 2018; Volume 12, Issue 6(Suppl3).
- Nguyen T, Dimov V, Bewtra A. Seminal fluid hypersensitivity in a man with postorgasmic illness syndrome (POIS). Annals of Allergy Asthma & Immunology. 2011; 107: A48.
- Serefoglu EC. Post-orgasmic Illness Syndrome: Where are we? J Sex Med. 2017 May; 14(5): 641-642.
- Nguyen HMT, Ahmed A, Gabrielson A, et al. Postorgasmic illness syndrome: A review, Sex Med Rev. 2018; 6: 11 – 15.
- Waldinger MD. Post orgasmic illness syndrome (POIS). Trans Androl Urol. 2016; 5(4): 602-606.
- Kim T, Shim Y, Lee S. Intralymphatic immunotherapy with autologous semen in a Korean man with Post-orgasmic illness syndrome, Sex Med Rev. 2018; 6: 174 – 179.
- Leppan H, Caulfiel A. Orgasmic migraine aura: Report of two cases, Cephalalgia. 2018; 1-4.
- Mashal A, Beer S. Post orgasmic illness: case report, J Sex Med. 2016; 13: 172-176.
- Depreux N, Basaga M, Pascalb M. Negative allergy study in a case of postorgasmic illness syndrome (POIS), Rev Int Androl. 2018; 16(1): 42-44.
- Bignami B, Honore T, Turmel N. Post orgasmic illness syndrome, Progrès en urologie; 2017; 27: 446-449.
- Reinert AE, Simon JA. “Did you climax or are you just laughing at me?” Rare phenomena associated with orgasm. Sex Med.
